Collect Every Data PointFrom Every ParticipantAll In One Platform

12Clinical Trial TechnologiesIn One Platform

Conduct Your ResearchIn the ClinicIn the HomeAnd On-the-Go

Conduct Real-TimeClinical Trialsto See Your Data Anytime

Co-create YourStudy Designwith Patient andHCP Insights

Add Our Experts toSelect and Develop YourClinical Trial Endpoints
Reduce YourResearch StudyCosts and Timeline
Explore Our No-Code Clinical Research Platform
Experience that Delivers Expertise for Our clients
Projects
+
Participants
+
Countries
+
Languages
+
Publications
+

Fully Configurable for Fit-For-Purpose Use
Our uniquely combined clinical research technology and consulting services help clients to design, operate, and scale next-generation research studies and electronic clinical outcome assessment (eCOA) programs for participants, sites, and study teams.
Measure What Matters Most
Grounded in the patient experience, we design meaningful outcome measures to optimize decision-making about the development, commercialization and use of medical products.
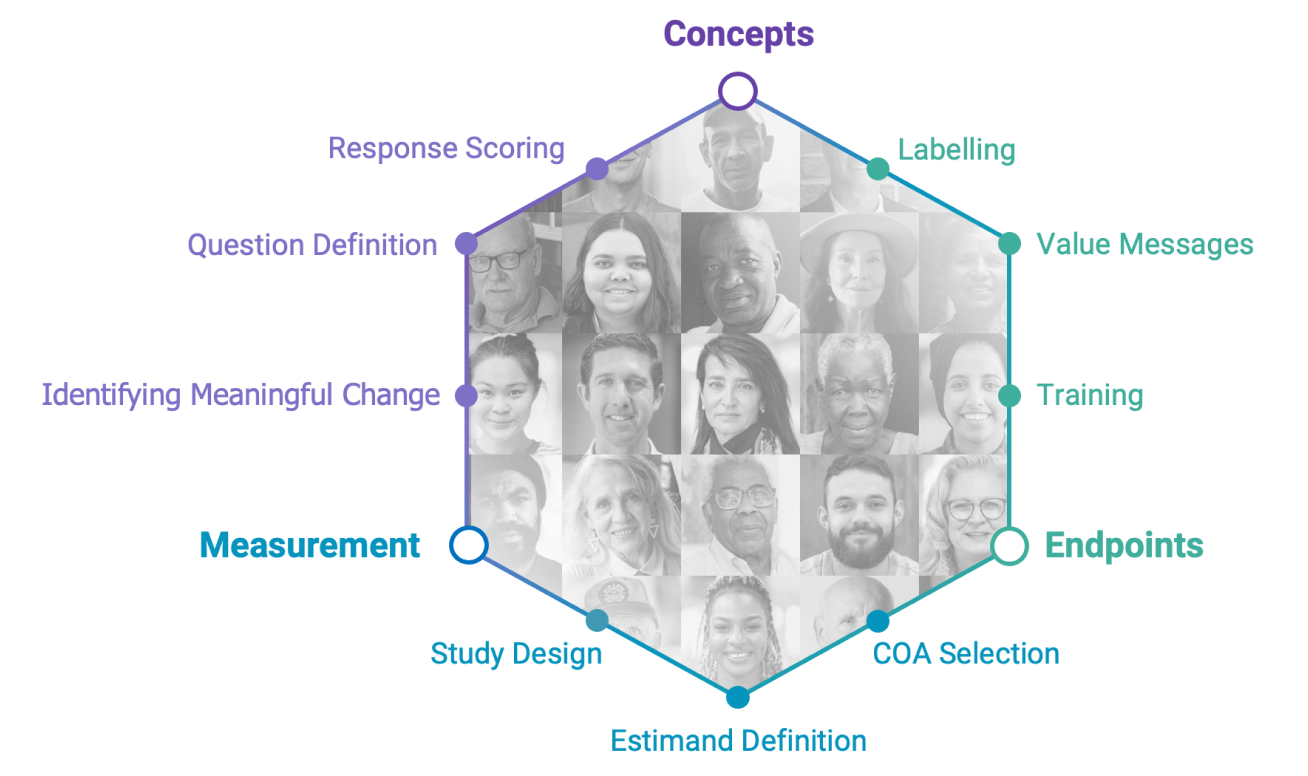

Listening to the Voice of the Patient and HCPs
Study sponsors and CROs understand the vital importance of listening to patients and caregivers throughout the drug development lifecycle. inVibe has solved those challenges with a methodology and technology to collect and analyze real patient voices that are simple, systematic, and scalable across the organization.
Next Generation of Research for Everyone, Everywhere
THREAD delivers one of the most trusted, patient-centric, comprehensive and advanced technologies in the industry. Our technology was built from the ground up to meet the needs of modern clinical trials — whether they be site-based, fully decentralized clinical trials (DCTs), or hybrid studies.








