Optimize Consent in Your Study
THREAD Delivers Compliant, Efficient eConsent to Reduce Your Site Burden
Secure Data Exchange
Send and receive participant consent with the flexibility to add features such as disclaimers for added data verification.
Smarter Compliance
Both single and dual signature eConsent / eAssist solutions are available with full compliance, audit trails, and reporting.
Mobile-Based
Participants need flexibility. eConsent is accessible through iOS or Android and supported in both BYOD and provisioned device approaches.
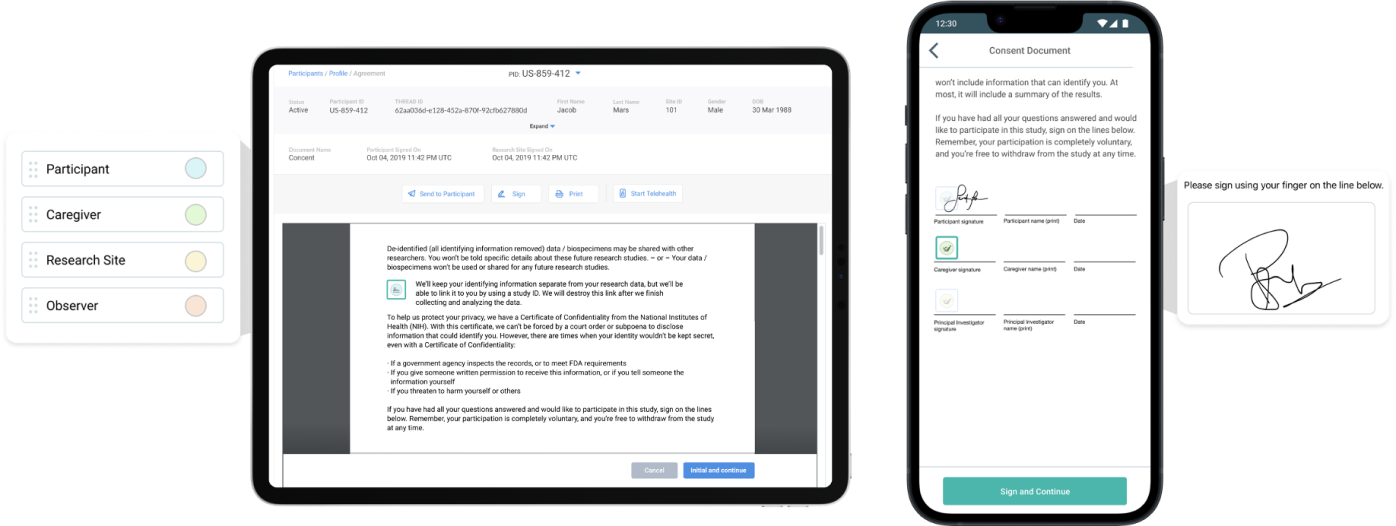
eConsent With Single Signature
Easy to use participant app includes education comprehension quiz, Informed Consent (ICF), and the ability to opt-in to health data access.
eConsent With Dual Signature(s)
Site teams and Principal Investigators (PIs) use THREAD to send ICFs or other study documents for signature to the participant remotely during a telehealth visit, phone call, or an in-clinic visit. The participant receives the document, reviews it in real time with the PI, and provides their digital signature. Immediately after the participant signs, the document is available for the PI to countersign and return the digital copy back to the participant.