
Reach Everyone, Everywhere
Next Generation Research Solution Built to Engage Patients
THREAD delivers one of the most trusted, patient-centric, comprehensive and advanced technologies in the industry. Our technology was built from the ground up to meet the needs of modern clinical trials — whether they be site-based, fully decentralized clinical trials (DCTs), or hybrid studies. We work hand in hand with our sponsor partners every step of the way, from protocol design all the way through launch, helping them to identify the most efficient and effective ways to run their studies.

Configurable Instrument Library for Rapid Deployment
Scientific Consulting Strategies to Create, Modify, Select, and Analyze Using insights from understanding the patient, endpoints can be configured and tested for confidence in viability.
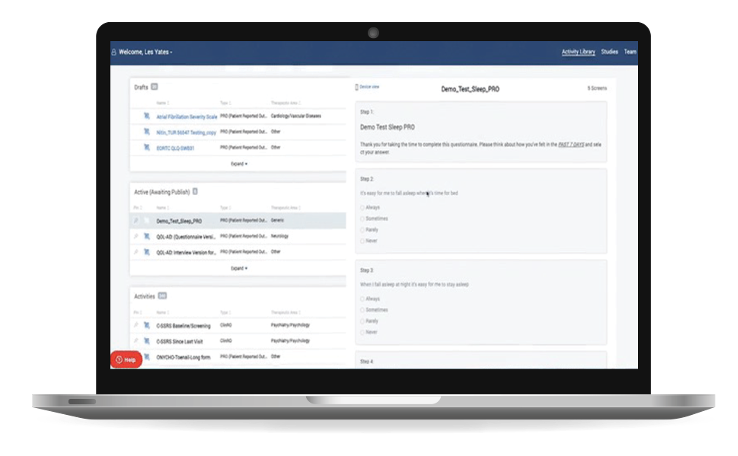
- Comprehensive eCOA Library
- Our 545+ eCOAs within the THREAD Library are pre-vetted, tested, and streamlined for licensing procurement
- Customize eCOA with a visual no-code editor
- Select and schedule any eCOA as needed for your study
Delivered on an Industry Leading Platform Built to Scale
Design
- Measure what matters with PCOR
- Capture Voice of the Patient, increase diversity in trials & optimize design
- Principles focused on Endpoint outcomes & optimized usability
Operate
- Operational design built around holistic patient journey for all studies
- World class professional services SMEs with deep industry experience
- Global instrument library validated instruments
Scale
- Omni-channel experience to keep sites and patients informed and engaged
- Ability to host data in regions to comply with global regulations
- Compliance engine to drive study health
- Measure - Study Health Dashboards & KPIs
- Respond - Direct to Patient notifications
- Adapt - Modern practices that eliminate waste & optimize value of no code
Reaching Everyone, Everywhere
Compliant and Secure — THREAD has the power to scale their studies however they like, enabling both full and hybrid DCTs models effectively.
- Flexibility to offer a broader range of activities — scheduled, unscheduled, or on-demand
- Simple, accessible data monitoring, all in one place
- Offline data capture ensures protocol compliance regardless of connectivity interruptions
- Omni-Channel Experience for Effective eCOA Deployment and Engagement

