Tracking Enrollment & Retention Data in Decentralized Clinical Trials
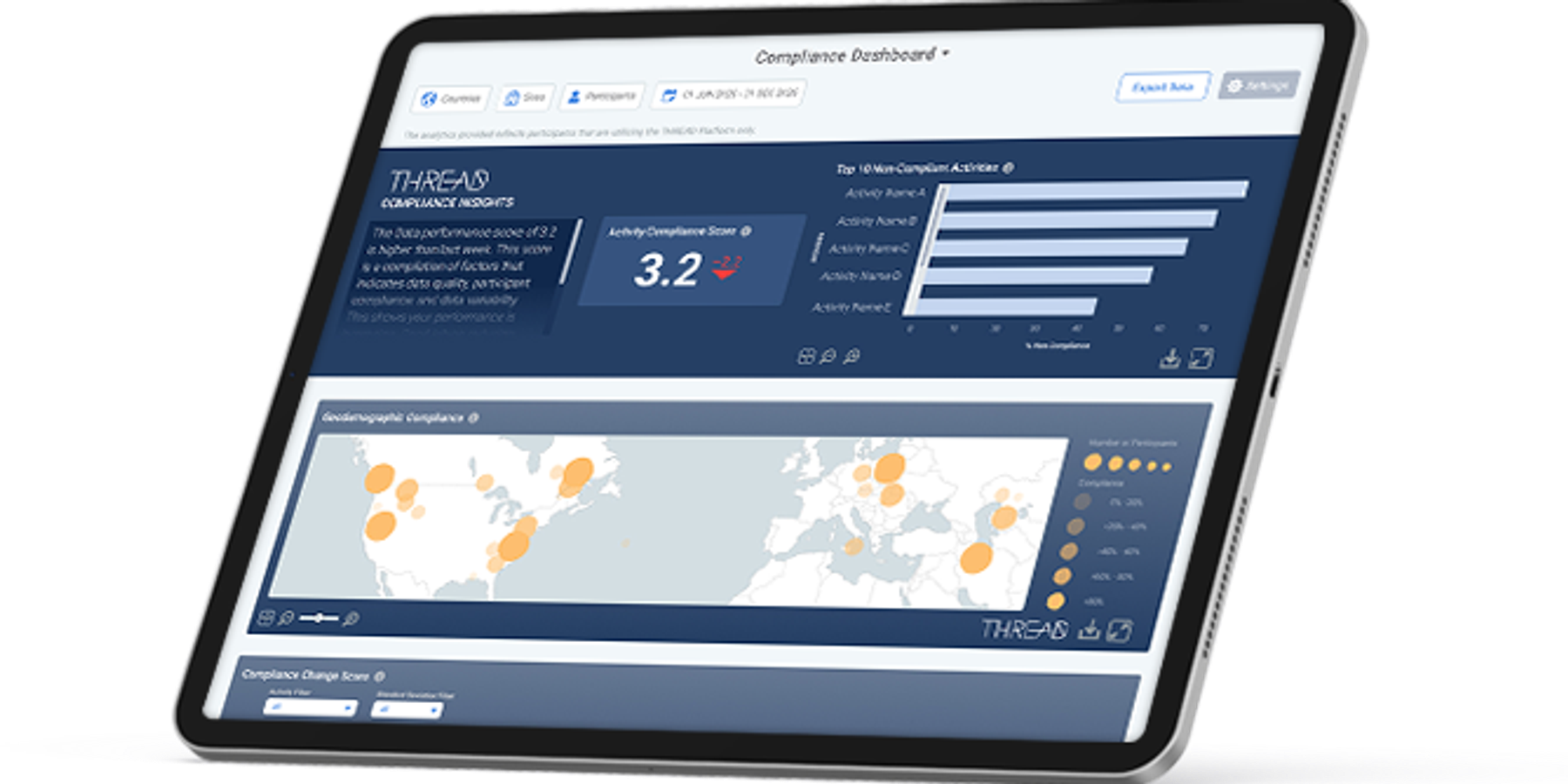
In our previous blog post on THREAD Analytics, we talked about the immediate need for more advanced performance metrics for today’s modern, decentralized clinical trial (DCT) approaches. These types of trials promise numerous benefits for all study stakeholders – sponsors, CROS, participants, etc. – in all phases of study execution. Arguably, the most important element of any clinical trial is participant enrollment and retention. After all, there can be no data without the people enrolled in the study.
DCTs offer many advantages related to enrollment and retention that traditional, site-based studies cannot match. By minimizing or eliminating the need for participants to visit clinical trial sites, we can greatly expand the geographic reach of a study. Additionally, the ability for remote technologies to provide always-on support and easy-to-use channels for engagement between participants and clinical teams can reduce stress and burden for participants and improve overall retention.
Advanced Metrics for Advanced Trial Enrollment
To fully realize the enrollment and retention benefits of DCT approaches, we need to be sure we are monitoring and measuring these studies effectively. In traditional studies, enrollment and retention metrics are calculated as the percentage of enrolled subjects who remain in a study without voluntarily withdrawing(1) and can be segmented by site, region, country – or any number of similar categories. Unfortunately, data updates in traditional studies are usually performed manually and often infrequently. They can also be impacted by site-specific problems, such as insufficient training or clinical team turnover.
THREAD Analytics, as we noted in our last blog post, are purpose-built for DCTs and hybrid clinical trials. They were developed to address the needs of real-time, continuous data submissions made possible by DCT approaches. This is because the usefulness of real-time data is largely lost without the ability to analyze that data as close to real-time as possible. Doing so allows clinical teams to effectively track enrollment and retention while receiving additional insights and details, including predictive trend data. Here is a look into how THREAD’s Enrollment/Retention Dashboard works:
- Data is submitted from a variety of sources (ePRO, ClinRO, telehealth visits, etc.)
- Data is analyzed within the THREAD platform to produce a proprietary THREAD score, along with other visualization tools (e.g., charts)
- The score provides immediate and real-time insight into increasing or decreasing trends over time alerting the user to developing issues
- THREAD’s proprietary algorithms power predictive analytics that is used to identify and show trends in enrollment and retention
Example: THREAD Enrollment/Retention Score
Example: A visualization of enrollment progress with trend data
Changes up or down in the score can signify many different things to study teams, such as the need for protocol changes, the need for additional training, too-high burden for the patient, etc. Real-time, comprehensive visibility into these trends helps study teams to identify problems quickly and make adjustments, as necessary.
Conclusion
Effective clinical trial enrollment and participant retention are critical to bringing new therapies to market. Recruitment, overall, represents as much as 40%(2) of clinical research budgets, and DCTs offer an avenue for introducing new efficiencies that can help to optimize enrollment and make studies run more smoothly. DCTs facilitate frequent and, in some cases, continuous submission of participant data. Old-school methods for study monitoring and management cannot accurately account for this kind of data flow. More importantly, they cannot mine the true value of real-time data. If we wish to fully leverage the advantages of DCT and hybrid trial approaches to optimize enrollment and retention, we must adopt methods for monitoring and measuring DCT recruitment data that are tailored specifically to these modern studies.
For more information on THREAD Analytics, download our report.
(1) Sullivan, L. “MCC Metric of the Month Blog: Subject Retention.” Applied Clinical Trials. May 2014.
(2) https://www. antidote.me/blog/25-useful-clinical-trial-recruitment-statistics-for-better-results
Matt Carson
Coordinator, THREAD
Matt Carson is a coordinator on the Data and Analytics team, leading the development of the THREAD Analytics dashboard.
He is a recent graduate from UNC with a Master's degree in Data Science.